- Blog
- Xtocc utility
- Cloud gardens
- 2018 cincinnati comic expo promo code
- Risk of rain 2 multiplayer
- White pages search
- Backblaze raises subscription pricing backup
- Bose usb audio driver windows 10
- Statplus regression wont pick up dependent variable
- The alan parsons project eve songs
- Driverpack solution 16 offline
- Cheap fifa 16 pc download
- Story planner for writers apk free
- Electron configuration for antimony
- Ark survival evolved servers
- Rimworld ideology conversion

Compounds taking up the other half are oxides, sulfides, sodium antimonate, and antimony trichloride. Batteries, antifriction alloys, type metal, small arms and tracer bullets, cable sheathing, and minor products use about half the metal produced. It greatly increases the hardness and mechanical strength of lead. UsesĪntimony is finding use in semiconductor technology for making infrared detectors, diodes and Hall-effect devices.

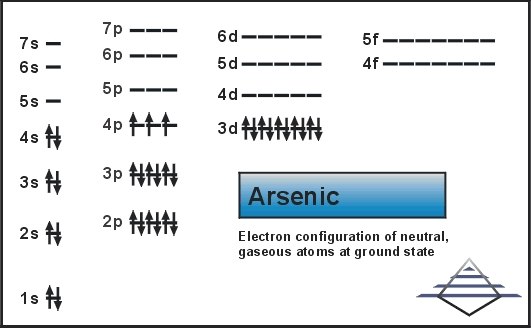
Antimony is a chemical element with the symbol Sb (from Latin: stibium) and atomic number 51. Antimony and many of its compounds are toxic. Electron configuration 4d 10 5s 2 5p 3: Electrons per shell: 2, 8, 18, 18, 5: Physical properties. It is a poor conductor of heat and electricity. It is sometimes found native, but more frequently as the sulfide stibnite. For example, 1s22s22p would be entered as 1s22s22p2. Express your answer in complete form, in order of increasing energy. Antimony was recognized in compounds by the ancients and was known as a metal at the beginning of the 17th. What electronic configuration corresponds to Antimony Sb +3. Electron configuration of Antimony is Kr 4d10 5s2 5p3. It is not abundant, but is found in over 100 mineral species. Transcribed image text: Enter the complete electron configuration for antimony. In the periodic table, the elements are listed in order of increasing atomic number Z.
#Electron configuration for antimony plus#
anti plus monos - a metal not found alone) Antimony was recognized in compounds by the ancients and was known as a metal at the beginning of the 17th century and possibly much earlier.
